KEY FEATURES
| Full Name | Cytochrome P450 2E1 |
|---|---|
| Synonym | CPE1; CYPIIE1; P450-J; P450C2E; 4-nitrophenol 2-hydroxylase; Cytochrome P450 Family 2 Subfamily E 1; Cytochrome P450,Subfamily IIE(Ethanol-Inducible)Polypeptide 1 |
| Assay Type | Sandwich |
| Reactivity | Mouse |
| Range | 3.12-200ng/mL |
| Sensitivity | 1.31ng/mL |
| Sample Type | Serum,plasma and other biological fluids |
| Sample Volume | 100μL |
| Detection Wavelength | OD450 |
| Transportation Temperature | 2-8°C |
| Specificity | The kit detected Mouse CYP2E1 in the samples and no significant cross-species reactions were found |
| Microplate | 96-wells plate breakable into 12 x 8 wells strip |
TEST PRINCIPLE
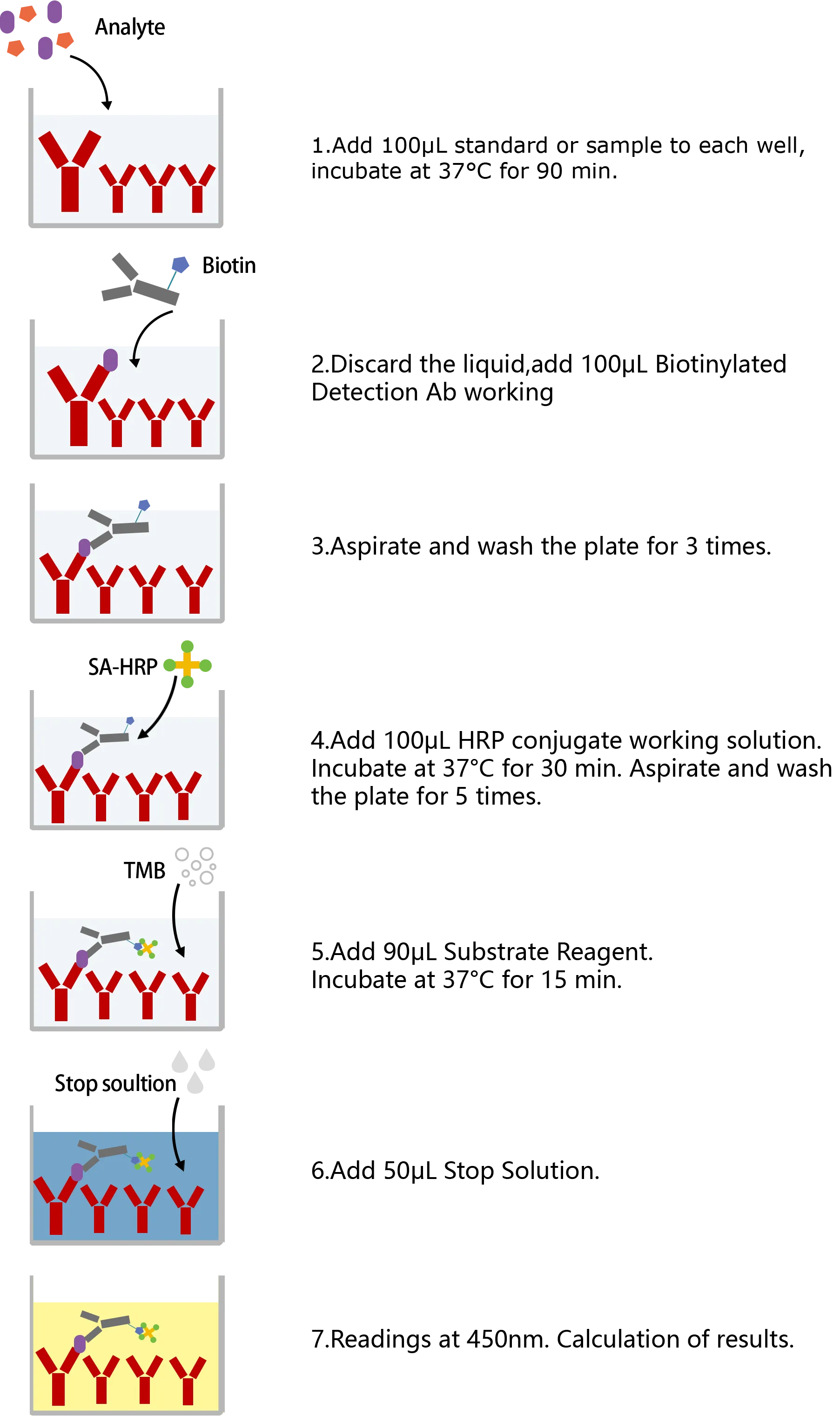
| This ELISA kit uses the Sandwich-ELISA principle. The micro ELISA plate provided in this kit has been pre-coated with an antibody specific to Mouse CYP2E1. Standards or samples are added to the micro ELISA plate wells and combined with the specific antibody. Then a biotinylated detection antibody specific for Mouse CYP2E1 and Avidin-Horseradish Peroxidase (HRP) conjugate are added successively to each micro plate well and incubated. Free components are washed away. The substrate solution is added to each well. Only those wells that contain Mouse CYP2E1, biotinylated detection antibody and Avidin-HRP conjugate will appear blue in color. The enzyme-substrate reaction is terminated by the addition of stop solution and the color turns yellow. The optical density (OD) is measured spectrophotometrically at a wavelength of 450 nm ± 2 nm. The OD value is proportional to the concentration of Mouse CYP2E1. You can calculate the concentration of Mouse CYP2E1 in the samples by comparing the OD of the samples to the standard curve. |
ELISA KIT COMPONENTS
Upon receipt, unpack promptly and store as recommended in the instructions.
| Components | Specifications | Storage and Notes |
|---|---|---|
| Micro Plate | 96T: 8 wells×12 strips 48T: 8 wells×6 strips | Unopened: -20°C, 12 months Unused: Put it back in the aluminum foil bag and seal it, store it at -20°C. |
| Reference Standard | 96T: 2 vials 48T: 1 vial | Unopened: -20°C, 12 months Please use freshly dissolved standards for each experiment. Discard any unused standards after dissolution. |
| Biotinylated Detection Ab Concentrate (100×) | 96T: 120μL×1 vial 48T: 60μL×1 vial | Unopened: -20°C, 12 months Unused: Please seal the concentrate and store it at -20°C, and discard the working solution. |
| HRP Conjugate Concentrate (100×) | 96T: 120μL×1 vial 48T: 60μL×1 vial | Unopened: -20°C(Protect from light), 12 months Unused: Please seal the concentrate and store it at -20°C, and discard the working solution. |
| Biotinylated Detection Ab Diluent | 14mL×1 | 2-8℃, 12 months |
| HRP Conjugate Diluent | 14mL×1 | 2-8℃, 12 months |
| Reference Standard & Sample Diluent | 20mL×1 | 2-8℃, 12 months |
| Washing Buffer Concentrate (25×) | 30mL×1 | 2-8℃, 12 months |
| Substrate Reagent(TMB) | 10mL×1 | 2-8°C(Protect from light),12 months |
| Stop Solution | 7mL×1 | 2-8°C/Room temperature |
ASSAY PROCEDURES
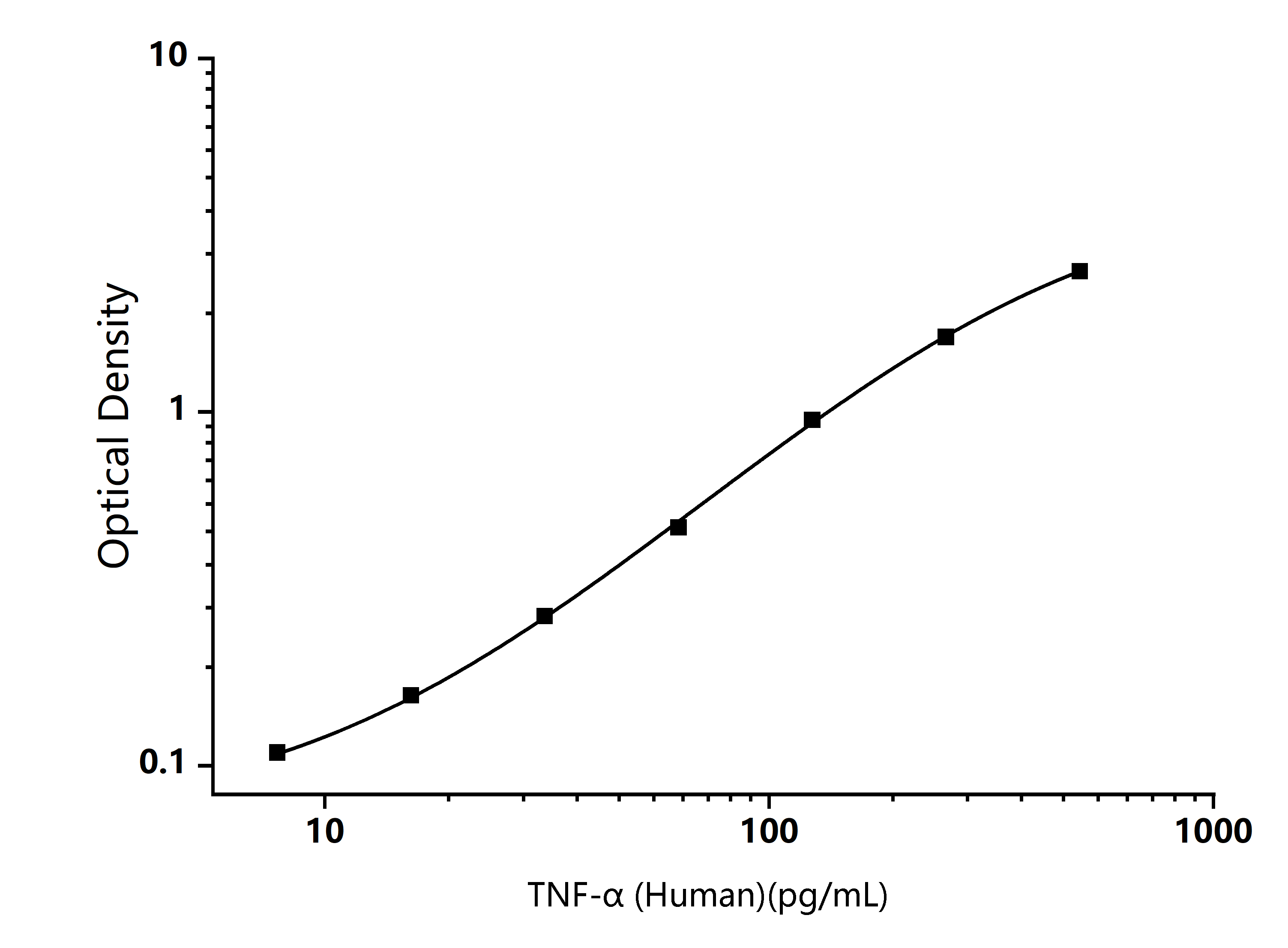
TYPICAL DATA
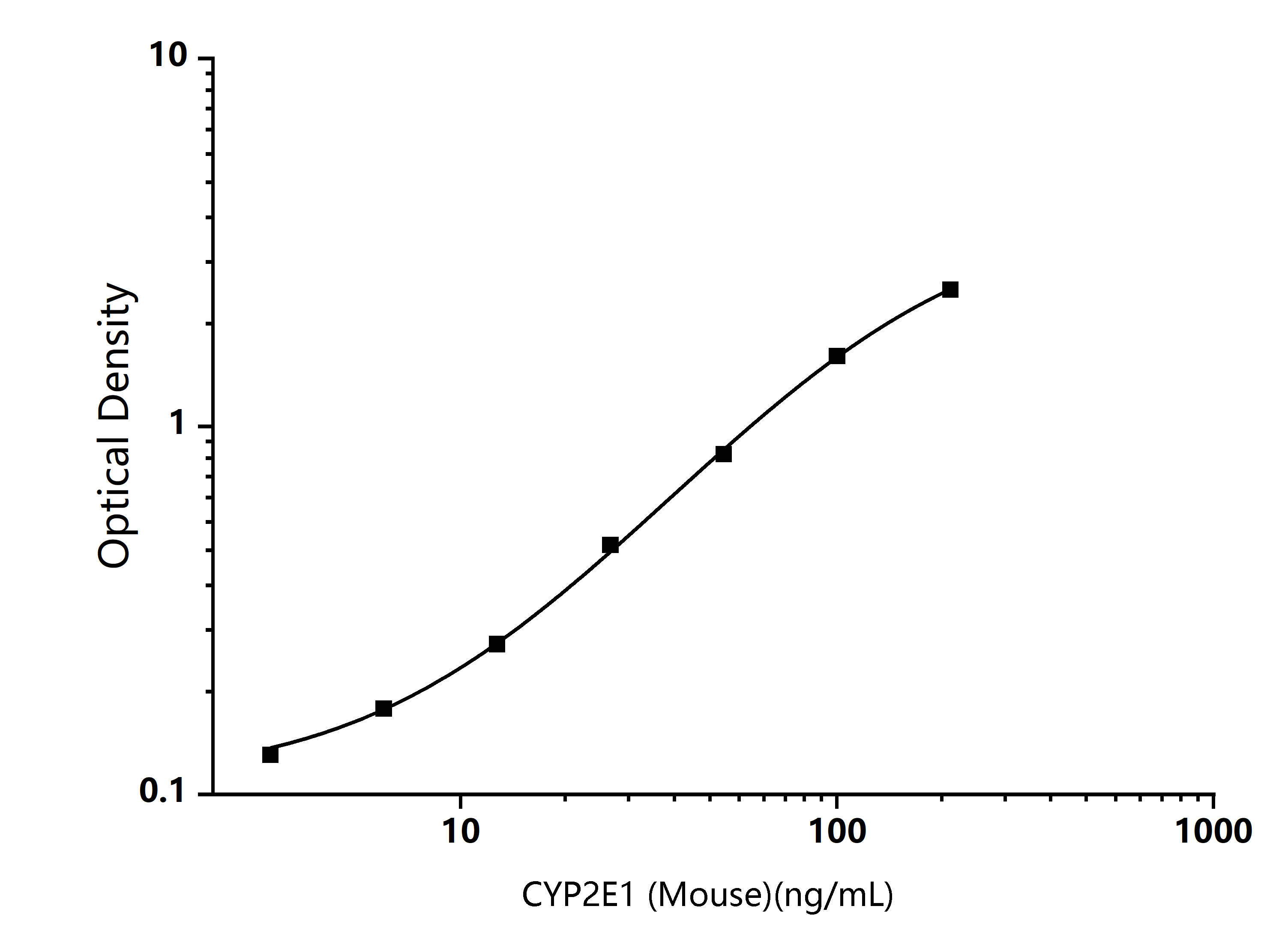
Mouse CYP2E1 ELISA Standard Curve
Typical data are for reference only and curves should be replotted for each experiment. The Logistics function is recommended for fitting.
PRECISION
Intra-Assay Precision (Precision within an assay): Three samples of known concentration were tested twenty times on one plate to assess intra-assay precision.
Inter-Assay Precision (Precision between assays): Three samples of known concentration were tested in twenty separate assays to assess inter-assay precision. Assays were performed by at least three technicians using two lots of components.
| Intra-assay Precision | Inter-assay Precision | |||||
|---|---|---|---|---|---|---|
| Sample | 1 | 2 | 3 | 1 | 2 | 3 |
| n | 20 | 20 | 20 | 20 | 20 | 20 |
| Mean (ng/mL) | 9.8 | 19.24 | 72.72 | 10.36 | 20.25 | 73.63 |
| Standard deviation | 0.57 | 0.82 | 3.16 | 0.7 | 1.19 | 2.87 |
| CV(%) | 5.85 | 4.24 | 4.34 | 6.76 | 5.88 | 3.9 |
RECOVERY
The recovery of Mouse CYP2E1 spiked to three different levels in samples throughout the range of the assay in various matrices was evaluated.
| Sample Type | Range (%) | Average Recovery (%) |
|---|---|---|
| Serum(n=8) | 84-98 | 90 |
| EDTA plasma (n=8) | 92-106 | 100 |
| Cell culture media (n=8) | 89-104 | 95 |
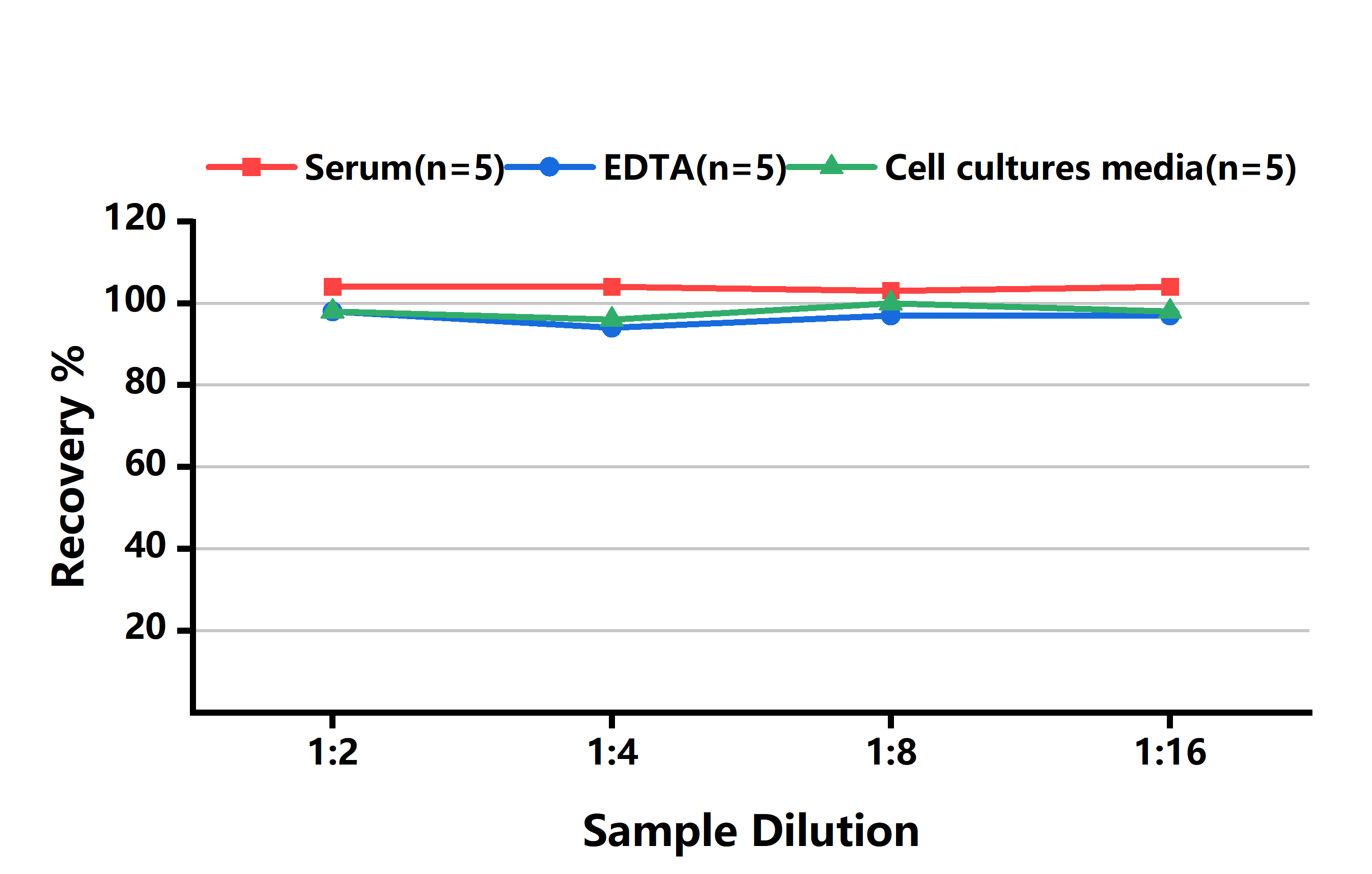
LINEARITY
To assess the linearity of the assay, samples containing and/or spiked with high concentrations of Mouse CYP2E1 in various matrices were diluted with the Reference Standard & Sample Diluent to produce samples with values within the dynamic range of the assay.