Comprehensive Guide to CCK-8 Assays: From Principle to Practice—Efficiently Obtaining Reliable Cell Proliferation Data
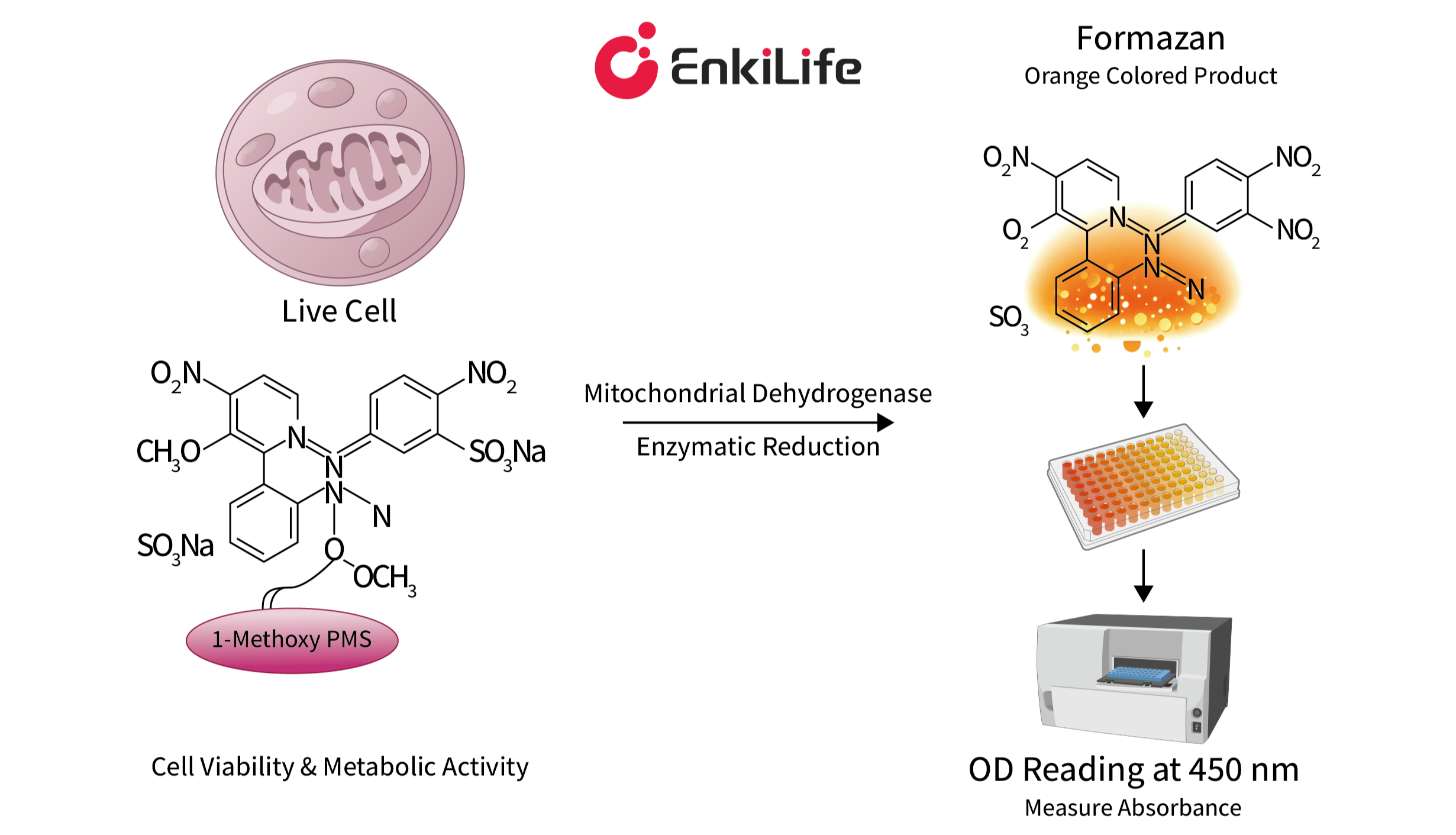
The reagent contains the water-soluble tetrazolium salt WST-8 [chemical name: 2-(2-methoxy-4-nitrophenyl)-3-(4-nitrophenyl)-5-(2,4-disulfophenyl)-2H-tetrazolium monosodium salt]. Under the mediation of an electron carrier—1-methoxy-5-methylphenazinium methylsulfate (1-Methoxy PMS)—this salt is reduced by mitochondrial dehydrogenases within viable cells into a highly water-soluble yellow formazan dye. The amount of formazan produced is directly proportional to the number of viable cells. By measuring the optical density (OD) value at a wavelength of 450 nm using a microplate reader, one can indirectly quantify the number of viable cells.