Literature Sharing: Multiplex Fluorescence Immunotechnology Reveals Colorectal Cancer Biomarkers
I. Research Background and Methods
Colorectal cancer (CRC) is a malignant tumor with high incidence and mortality worldwide, accounting for approximately 9.7% of global cancer cases and 8.5% of cancer deaths. Despite new research insights into the molecular pathogenesis of colorectal cancer, existing treatment methods still cannot significantly improve patients' overall survival. A considerable proportion of patients experience local recurrence and distant metastasis within 5 years after surgery, and even with multidisciplinary comprehensive diagnosis and treatment making some progress, patients' disease-free survival remains unsatisfactory. Meanwhile, the traditional TNM staging system based on tumor size, lymph node, and distant metastasis, combined with histological grading for disease risk stratification, is insufficient for accurate prognostic assessment of individual colorectal cancer patients. Currently used serological markers such as carcinoembryonic antigen (CEA) for diagnosis can detect disease recurrence after treatment, but due to marker changes not being detectable in all patients, there are issues with reliability. Therefore, there is an urgent clinical need to discover new clinicopathological and prognostic markers for colorectal cancer. To address this need, in the article "Multiplex Immunohistochemistry Indicates Biomarkers in Colorectal Cancer," the authors innovatively used multiplex fluorescence immunohistochemistry (mIHC) technology, using tumor tissue and adjacent normal tissue from 84 colorectal cancer patients to construct tissue microarrays (TMA), simultaneously detecting the expression levels of cancer stem cell marker CD133, immune checkpoint molecule PD-L1, tumor-associated antigen HER2, and macrophage marker CD68, combining clinicopathological features with 7-year follow-up data to explore the diagnostic and prognostic value of these markers in colorectal cancer, providing new technical support and theoretical basis for precise diagnosis and treatment of colorectal cancer.
In terms of research methods, the researchers used tissue microarrays, selected 84 colorectal cancer patient samples with complete clinical and follow-up data, optimized mIHC staining conditions, used the PANO 7-plex IHC kit for multiplex fluorescence staining of CD133, HER2, PD-L1, and CD68, obtained multispectral images using the Ploaris system, performed spectral unmixing and H-score quantitative analysis using InForm image analysis software, analyzed marker expression differences using Mann-Whitney U test, analyzed the association between markers and patients' overall survival (OS) using Kaplan-Meier method combined with log-rank test, and completed all statistical analyses using GraphPad Prism 8.0 software.
II. Core Technical Highlight: mIHC
Multiplex fluorescence immunohistochemistry is the core technology of this study. Compared with traditional single-label immunohistochemistry, its greatest advantage is the ability to simultaneously detect the expression levels of multiple target proteins on the same tissue section while preserving the spatial distribution relationship of each protein within the tissue. This effectively solves the limitation of the traditional technology's "single target per section" and avoids the interference of tissue heterogeneity between different sections, improving detection efficiency and result accuracy. By constructing a spectral library, optimizing antibody concentration and staining sequence, and combining TSA fluorescence labeling technology with DAPI nuclear staining, the researchers achieved clear labeling and precise quantification of four proteins: CD133, HER2, PD-L1, and CD68, laying a technical foundation for subsequent analysis of the expression association and clinical significance of the four markers, fully demonstrating the unique value of mIHC technology in combined detection of multiple tumor markers.
III. Research Result Analysis
Association Analysis between Clinicopathological Features and 7-Year Overall Survival
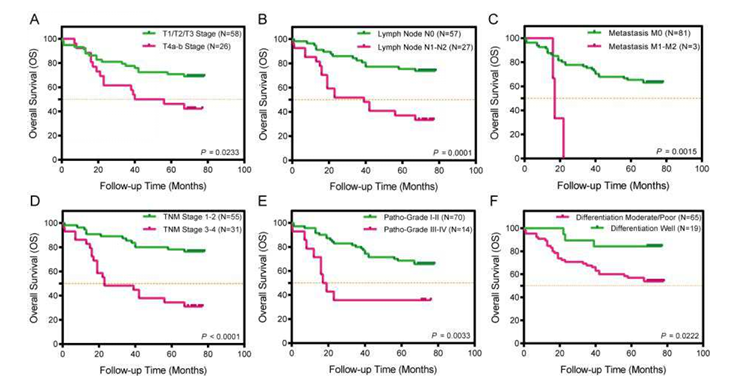
The Kaplan-Meier survival curve focuses on the 7-year overall survival (OS) of 84 colorectal cancer patients, systematically analyzing the prognostic value of 6 clinicopathological features: T stage, lymph node metastasis, distant metastasis, TNM stage, pathological grade, and differentiation degree. The orange dashed line in the figure represents 50% overall survival rate. Figure A shows T stage, B shows lymph node metastasis status, C shows distant metastasis status, D shows TNM stage, and E and F show pathological grades. This figure clearly demonstrates that the prognosis of colorectal cancer patients is closely related to the degree of tumor progression, providing a clinical basis for subsequent analysis of the association between biomarkers and prognosis.
mIHC Atlas of Colorectal Cancer Tissues
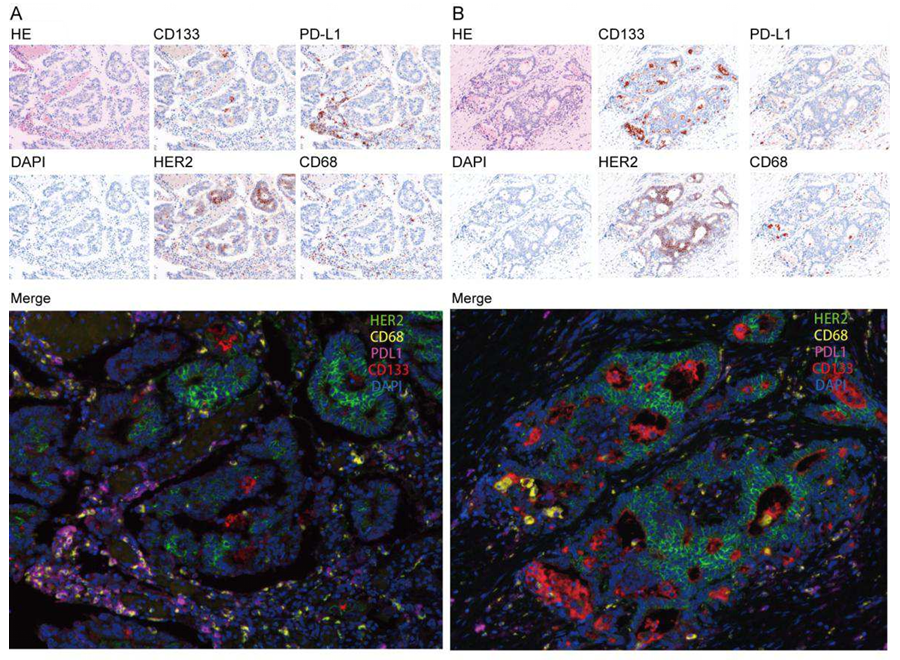
Focusing on the staining effects of mIHC technology in colorectal cancer tissues, representative images of two types of cancer tissues from the sigmoid colon (Figure A) and ascending colon (Figure B) clearly outline the distribution characteristics of single-label and multi-label fluorescence. The small images at the top and middle of the figure are HE staining, DAPI staining, and single-label fluorescence images of CD133, PD-L1, HER2, and CD68 in sequence, while the large image at the bottom is the multi-label fluorescence fusion image of the four proteins with DAPI. Experimental observations found: all four target proteins are expressed in cancer tissues to varying degrees, CD133 is mainly expressed on the surface of cancer cells, PD-L1 is mostly expressed on the surface of cancer cells and immune cells, HER2 is mainly localized on cancer cell membranes, and CD68 is mainly distributed in macrophages in the tumor microenvironment. The multi-label fusion images can clearly distinguish the spatial distribution of the four proteins, intuitively presenting the co-expression of each marker in the same tissue region, fully verifying the advantage of mIHC technology in achieving simultaneous detection of multiple targets on a single section, and providing reliable image basis for subsequent quantitative analysis.
Expression Difference Analysis of Each Marker and Their Combinations
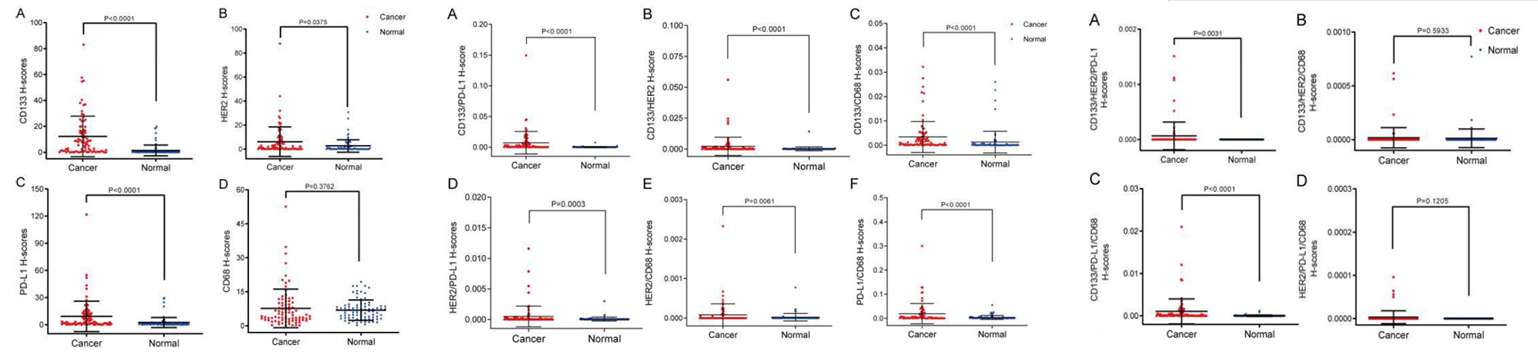
Combining core data on single-label marker expression differences, double-label marker combination expression differences, triple-label marker combination expression differences, and the association between markers and clinicopathological features, comprehensive analysis was conducted focusing on the single-label expression, combination expression characteristics of CD133, HER2, PD-L1, and CD68, and their association with clinicopathological features, to clarify the clinical value of each marker and combination.
From the perspective of single-label marker expression, using H-score as the quantitative indicator, CD133, HER2, and PD-L1 are significantly higher in colorectal cancer tissues than in adjacent normal tissues, showing tissue specificity and can be used as potential diagnostic markers. However, CD68 shows no significant difference in expression between the two types of tissues, and its role is related to tumor microenvironment regulation.
In terms of marker combinations, all six double-label combinations show significantly higher expression in cancer tissues than in normal tissues, with the most significant differences in the combinations of CD133 with PD-L1, HER2, CD68, and PD-L1 with CD68, suggesting synergistic expression relationships between markers. Among the four triple-label combinations, only CD133/HER2/PD-L1 and CD133/HER2/CD68 show significant expression differences, indicating that combinations centered on CD133 and HER2 have greater diagnostic potential.
Association Analysis between Markers and Clinicopathological Features and Patient Prognosis
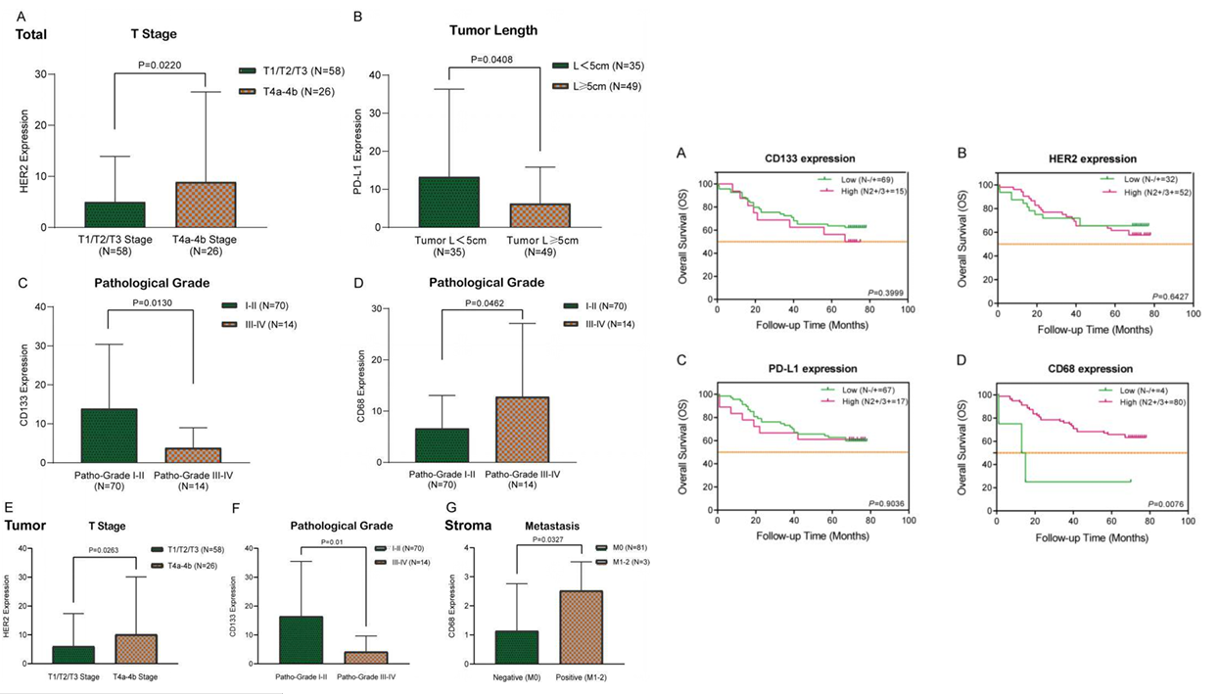
Association with clinicopathological features shows that HER2 expression is higher in T4a-b stage than in T1-T3 stage, which can indicate tumor progression. PD-L1 expression is higher in tumors with maximum diameter ≥5cm, CD133 expression is higher in grade I-II pathological grades, and CD68 expression is higher in grade III-IV and those with distant metastasis, confirming its role in tumor microenvironment and metastasis. Combined with Kaplan-Meier survival curve analysis, the four markers were divided into low and high expression groups based on H-score. The results showed no significant difference in survival rates between low and high expression groups for CD133, HER2, and PD-L1, only the high CD68 expression group had significantly higher survival rate than the low expression group. In conclusion, CD133, HER2, and PD-L1 are mainly related to tumor clinicopathological features and can assist in disease assessment, while CD68 not only participates in tumor microenvironment regulation and promotes metastasis but also is closely related to patient prognosis. High CD68 expression indicates better prognosis, and the four markers together provide multi-dimensional references for precise diagnosis, disease assessment, and prognostic judgment of colorectal cancer.
IV. Conclusion
Through the precise application of mIHC, this study clarified the expression characteristics and clinical significance of four markers (CD133, HER2, PD-L1, and CD68) in colorectal cancer tissues, namely CD133 and PD-L1 can serve as potential biomarkers for early diagnosis of colorectal cancer, HER2 can serve as an indicator for judging tumor progression, and CD68 is not only related to tumor metastasis but also can serve as a potential marker for prognostic assessment. This study fully demonstrates the advantages of mIHC technology in combined detection of multiple tumor markers, which can achieve simultaneous detection and spatial localization of multiple targets on a single tissue section, providing an efficient and reliable technical method for precise diagnosis, prognostic assessment, and pathogenesis research of tumors.
In addition, this study also provides important insights for subsequent related research. In clinical diagnosis and treatment of colorectal cancer, mIHC technology can be used to construct a combined detection system of multiple markers to improve the precision of diagnosis and prognostic assessment. At the same time, the dual role of CD68 in colorectal cancer, which can promote metastasis and improve prognosis, also suggests that subsequent research needs to further explore the polarization mechanism of macrophages in the tumor microenvironment, providing a theoretical basis for the development of new immunotherapy targets. Overall, this study not only enriches the research results of colorectal cancer biomarkers but also promotes the application and development of mIHC technology in clinical tumor detection.
References
Zhang W, Song ZJ, Zhang BY, Wang JL, Guo Q, Sun ZW, Tang H. Multiplex immunohistochemistry indicates biomarkers in colorectal cancer. Neoplasma. 2021 Nov;68(6):1272-1282. doi: 10.4149/neo_2021_210312N324. Epub 2021 Aug 30. PMID: 34459208.
EnkiLife mIHC TSA Kits
Product | Catalog Number |
|---|---|
TSA Six-Label Seven-Color Multiplex Immunohistochemistry Kit | |
TSA Five-Label Six-Color Multiplex Immunohistochemistry Kit | |
TSA Four-Label Five-Color Multiplex Immunohistochemistry Kit | |
TSA Three-Label Four-Color Multiplex Immunohistochemistry Kit | |
TSA Two-Label Three-Color Multiplex Immunohistochemistry Kit |
