——Efficiently Obtaining Reliable Cell Proliferation Data
The reagent contains the water-soluble tetrazolium salt WST-8 [chemical name: 2-(2-methoxy-4-nitrophenyl)-3-(4-nitrophenyl)-5-(2,4-disulfophenyl)-2H-tetrazolium monosodium salt]. Under the mediation of an electron carrier — 1-methoxy-5-methylphenazinium methylsulfate (1-Methoxy PMS) — this salt is reduced by mitochondrial dehydrogenases within viable cells into a highly water-soluble yellow formazan dye. The amount of formazan produced is directly proportional to the number of viable cells. By measuring the optical density (OD) value at a wavelength of 450 nm using a microplate reader, one can indirectly quantify the number of viable cells.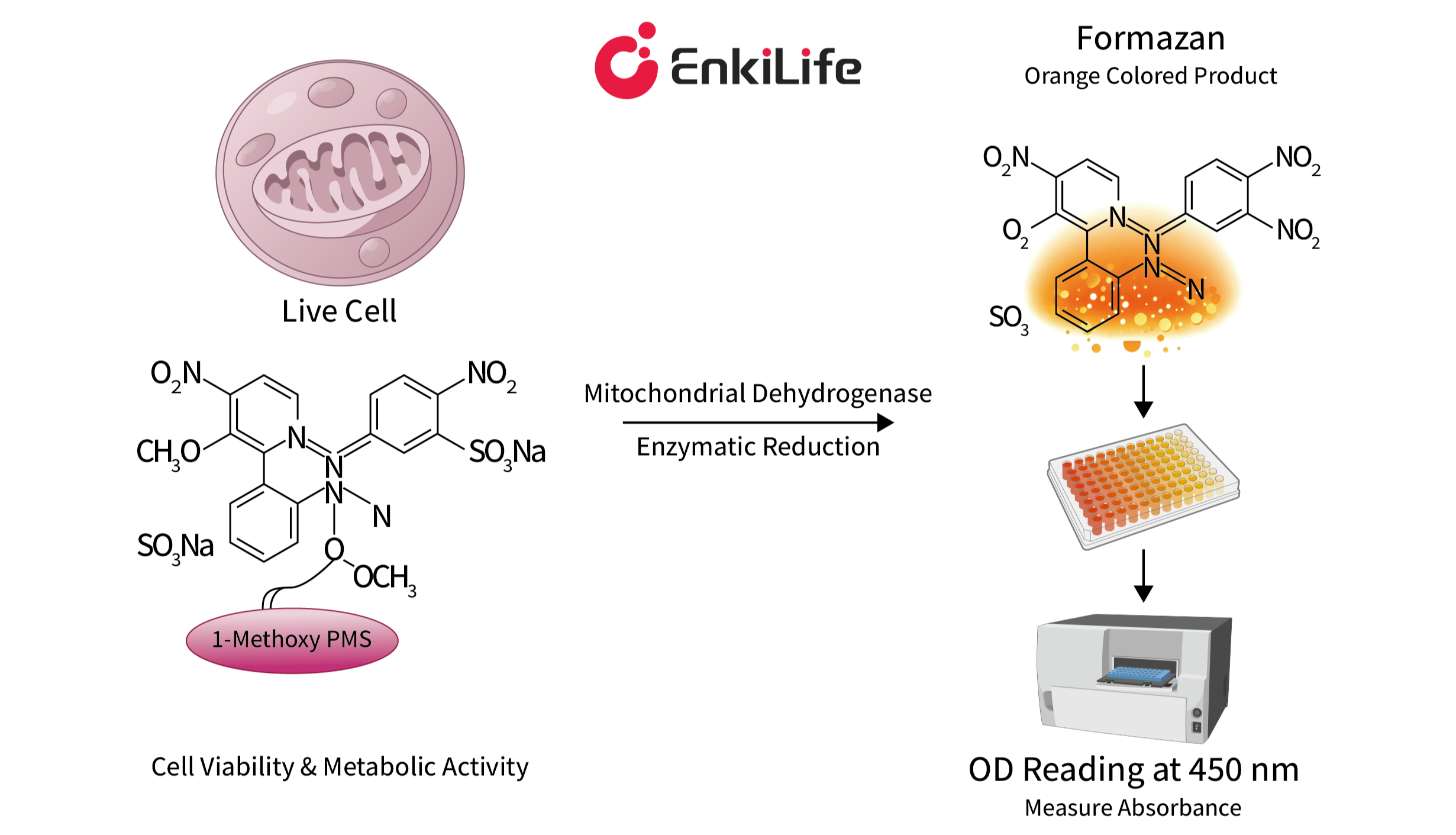
Schematic Diagram of the CCK-8 Assay Principle
- Cell viability detection
- Cytotoxicity detection
- Cell proliferation detection
- Drug screening
Experimental Group: Contains cells, culture medium, test drug, and CCK-8.
Control Group: Contains cells, culture medium, and CCK-8.
Blank Group: Contains culture medium and CCK-8.
Due to the "edge effect" — where humidity in the outermost wells is lower than in the central wells, leading to more severe water evaporation — the outermost row of wells in the 96-well plate should be left untreated (i.e., not used for samples); instead, add 200 µL of PBS or cell culture medium to these wells.
Each experimental group requires a minimum of three replicate wells.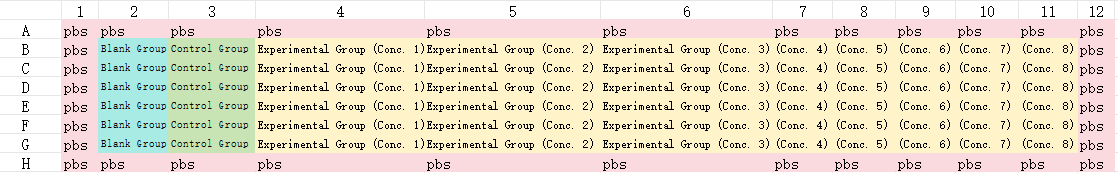
Seed 100 µL of cell suspension into each well of the 96-well plate at a density of 3,000–7,000 cells per well (use cells that are in good condition with a confluence of ≥80%). Continue to culture the cells for 24–48 hours under standard conditions: 37°C, 5% CO2, and 90% humidity.
Important Notes:
a. The specific cell density required will vary depending on the cell type, cell size, and proliferation rate.
b. When plating cells, ensure continuous mixing of the cell suspension while dispensing to maintain uniformity. It is highly recommended to use a multi-channel pipette to minimize the plating time. Immediately after plating, visually inspect the wells to verify that the cell density is uniform; if any wells exhibit uneven density, mark them and discard them.
Once the cells have adhered to the plate, aspirate the existing culture medium. Add drug-containing medium to the experimental groups, and add drug-free medium to the control groups. Subsequently, place the plate into a cell culture incubator for incubation.
Precautions:
a. Drug Concentration Selection: Establish a concentration gradient centered around the drug's IC50 value, spanning a specific range of both higher and lower concentrations. If the IC50 value for the drug is unavailable, conduct a preliminary experiment to determine an appropriate range. This preliminary experiment may begin with a broad-range screen — for instance, testing concentrations of 0.01 µM, 0.1 µM, 1 µM, 10 µM, and 100 µM (5 concentrations in total). Based on these results, narrow down the range to be used for the formal experiment. The goal is to identify concentrations approximating the drug's Minimum Inhibitory Concentration (MIC) and the concentration required for complete inhibition (100% inhibition).
b. For suspension cells, it is generally not necessary to aspirate the culture medium prior to drug addition; however, ensure that the final drug concentration within the well is accurate.
If each well contains 100 µL of culture medium, add 10 µL of CCK-8 reagent to each well (maintaining a ratio of CCK-8 to medium of 1:10). Place the plate into the incubator for incubation. Incubate for 1 to 4 hours; generally, a longer incubation time results in a higher OD (Optical Density) value.
Precautions: Ensure that the incubation time for assessing cell viability remains consistent across all time points!
Use a microplate reader to measure the absorbance at a wavelength of 450 nm (which serves as an indirect indicator of the number of viable cells).
Precautions: If immediate measurement is not possible, the reaction must be halted. This can be achieved by placing the plate in a refrigerator at 4°C; measurements should then be performed within 24 hours. If you require a precise determination of the actual cell count, you may construct a standard curve; the resulting OD values are typically considered reliable within the range of 1.0 to 2.0.
(1) The formula for calculating cell viability is as follows:
(2) The formula for calculating cell inhibition rate is as follows:
If you intend to calculate the IC50 value, you should calculate the inhibition rate. This is because the IC50 refers to the concentration of a drug required to inhibit a specific biological process by 50%. It reflects the drug's "inhibitory" effect rather than the cells' "survival" status.
This should be determined through preliminary experiments. Typically, for a 96-well plate, 3,000 to 7,000 cells are seeded per well (adjusted according to the cell growth rate) to ensure that, at the end of the culture period, the cells in the control wells have just reached full confluence or are nearly confluent (approximately 80–90% confluence).
It will have a significant impact. Ensure that the cells used for the experiment are in the logarithmic growth phase, possess high viability, and are in a stable condition; avoid using cells that have undergone too many passages.
Possible causes include: excessive trypsin digestion (leading to the disruption of intercellular junctions); insufficient serum concentration in the culture medium; or poor intrinsic cell condition (e.g., failure to fully recover after thawing).
Solutions: 1. Monitor the trypsin digestion process under a microscope; as soon as the intercellular spaces widen and the cell edges become rounded, immediately add serum-containing medium to terminate the digestion. 2. Adjust the serum concentration in the culture medium to 10–15%. 3. After thawing, culture the cells for 48 hours to allow them to fully adhere and spread before proceeding with drug treatment.
Yes, but the following steps are required: Carefully aspirate the culture medium containing the CCK-8 reagent and the orange-yellow formazan product (be careful not to aspirate the cells!). Gently rinse the wells 2–3 times with pre-warmed PBS to thoroughly wash away any residual reagent. Then, add complete culture medium and return the plate to the incubator.
The drug inhibits the activity of cellular metabolic enzymes (without causing cell death); competitive inhibition occurs between the CCK-8 reagent and the drug; or the cells have entered a dormant phase.
Solutions: 1. Verify cell viability using a live/dead cell staining kit (e.g., Calcein-AM/PI) to rule out interference from metabolic inhibition. 2. Include a "Drug + CCK-8 + Culture Medium" blank control group to subtract the drug's influence on color development. 3. Extend the recovery period following drug treatment (e.g., by culturing for an additional 12 hours) before measuring the OD values.
Air bubbles are present in the cell suspension (introduced during pipetting); impurities are present at the bottom of the culture plate wells (due to insufficient cleaning); or the cell suspension concentration is uneven.
Solutions: 1. Inspect the cell suspension before pipetting; if bubbles are present, use a pipette tip to puncture and eliminate them. 2. Rinse the culture plates twice with ultrapure water before use, and allow them to air-dry completely. 3. Thoroughly mix the cell suspension before aliquoting, and re-mix it after dispensing every 10 wells.
Reduce the drug concentration or shorten the treatment duration; alternatively, verify whether the toxicity is caused by the solvent (e.g., DMSO).
Solution: No. The working principle of CCK-8 relies on the dehydrogenase enzymes located within the mitochondria of eukaryotic cells.
Solution: Adherent cells undergo four distinct stages after seeding: the "suspension phase" (0–6 h), the "attachment phase" (6–12 h), the "spreading phase" (12–24 h), and the "proliferation phase" (after 24 h). The true effects of a drug can only be accurately reflected once the cells have completed attachment and spreading, and have entered the proliferation phase. Therefore, under standard experimental conditions, it is recommended to add the drug 24–48 hours after seeding. Unlike adherent cells, suspension cells do not require a waiting period to attach and adapt. Provided the seeding density is appropriate, they typically enter the logarithmic growth phase — characterized by vigorous metabolism, stable proliferation, and high sensitivity to drug responses — within 12–24 hours post-seeding. Therefore, under standard experimental conditions, it is recommended to add test compounds within 12–24 hours after seeding.
Typically, the incubation period is 1–4 hours. The optimal incubation time should be determined through preliminary experiments; the linear correlation is strongest when the OD values fall within the range of 1.0 to 2.0.
Product Details:Enhanced Cell Counting Kit-8
 | Flora Flora is a technical support expert at EnkiLife, familiar with immunology and neuroscience, dedicated to providing customers with high-quality product combinations and technical support to help achieve research in neurodegenerative diseases and other neuroscience areas. |
