Product Introduction
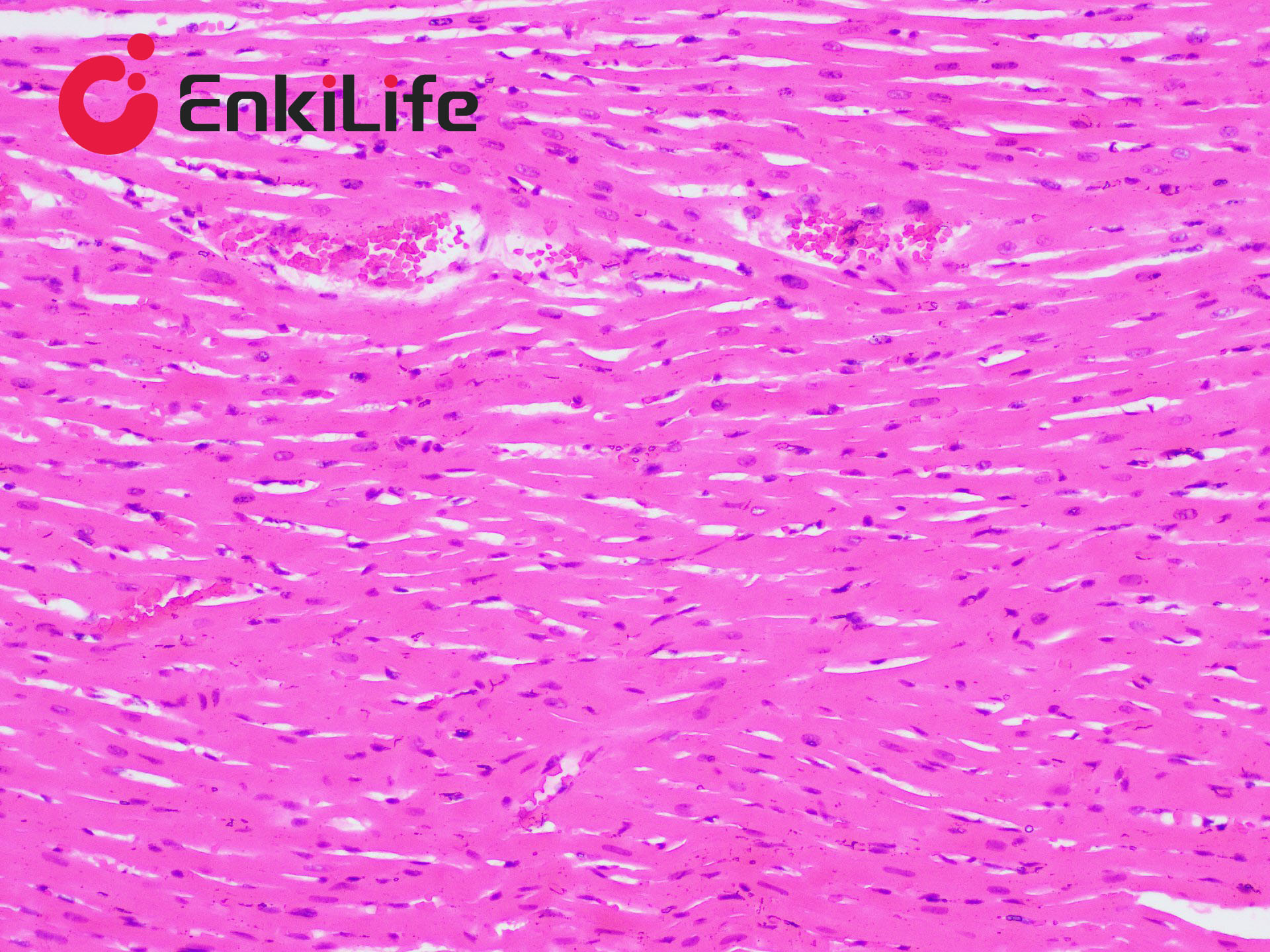
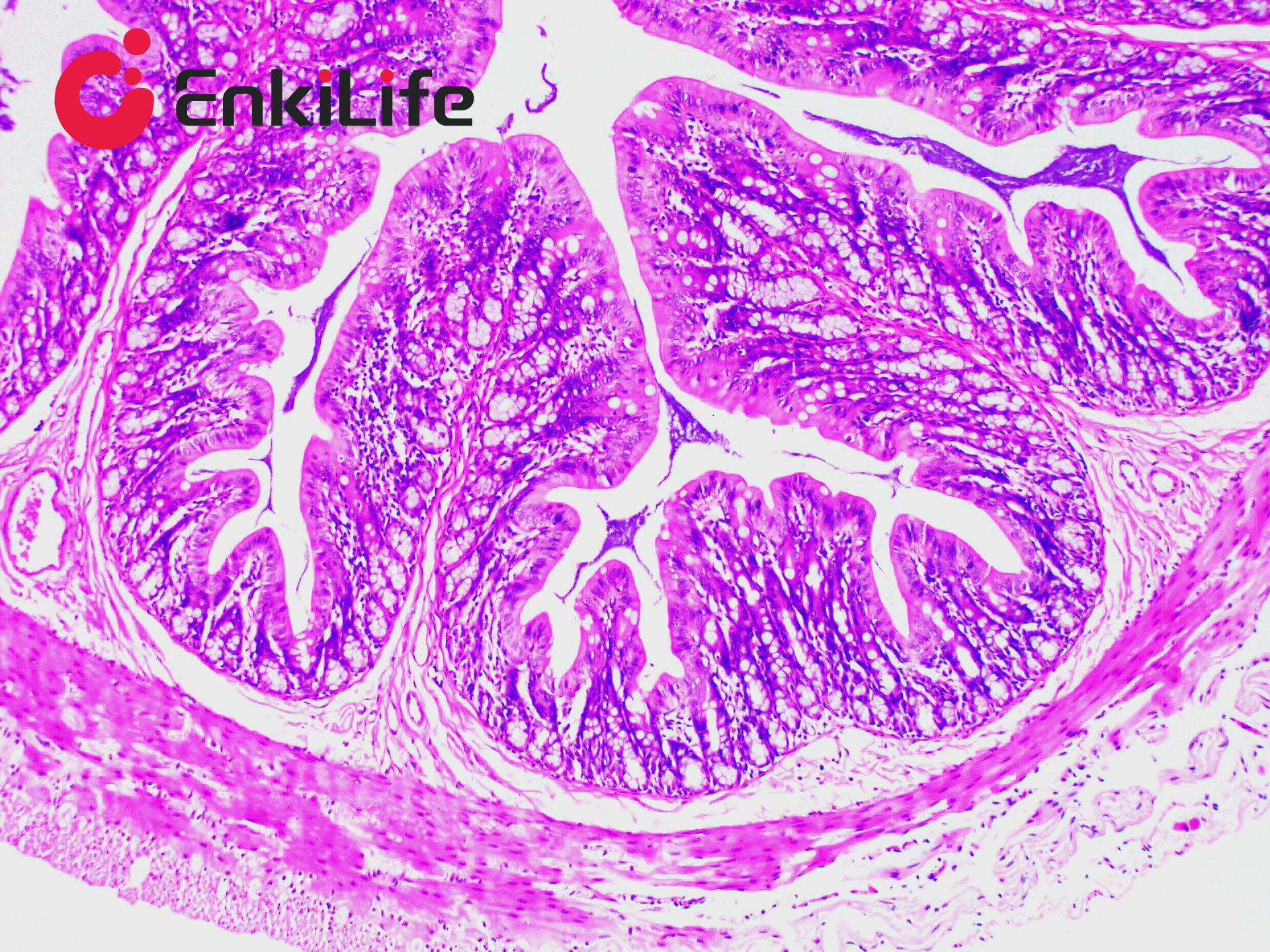
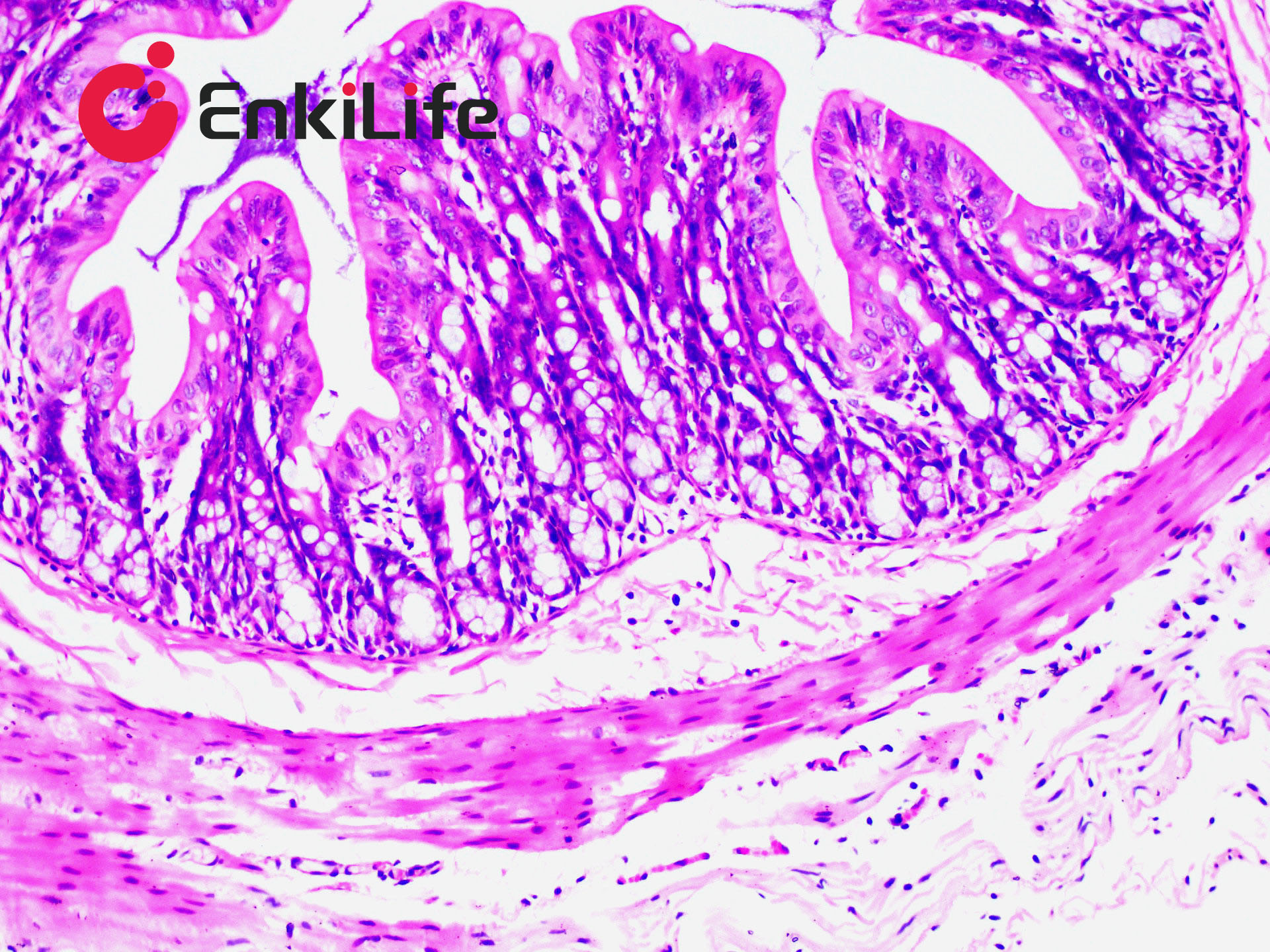
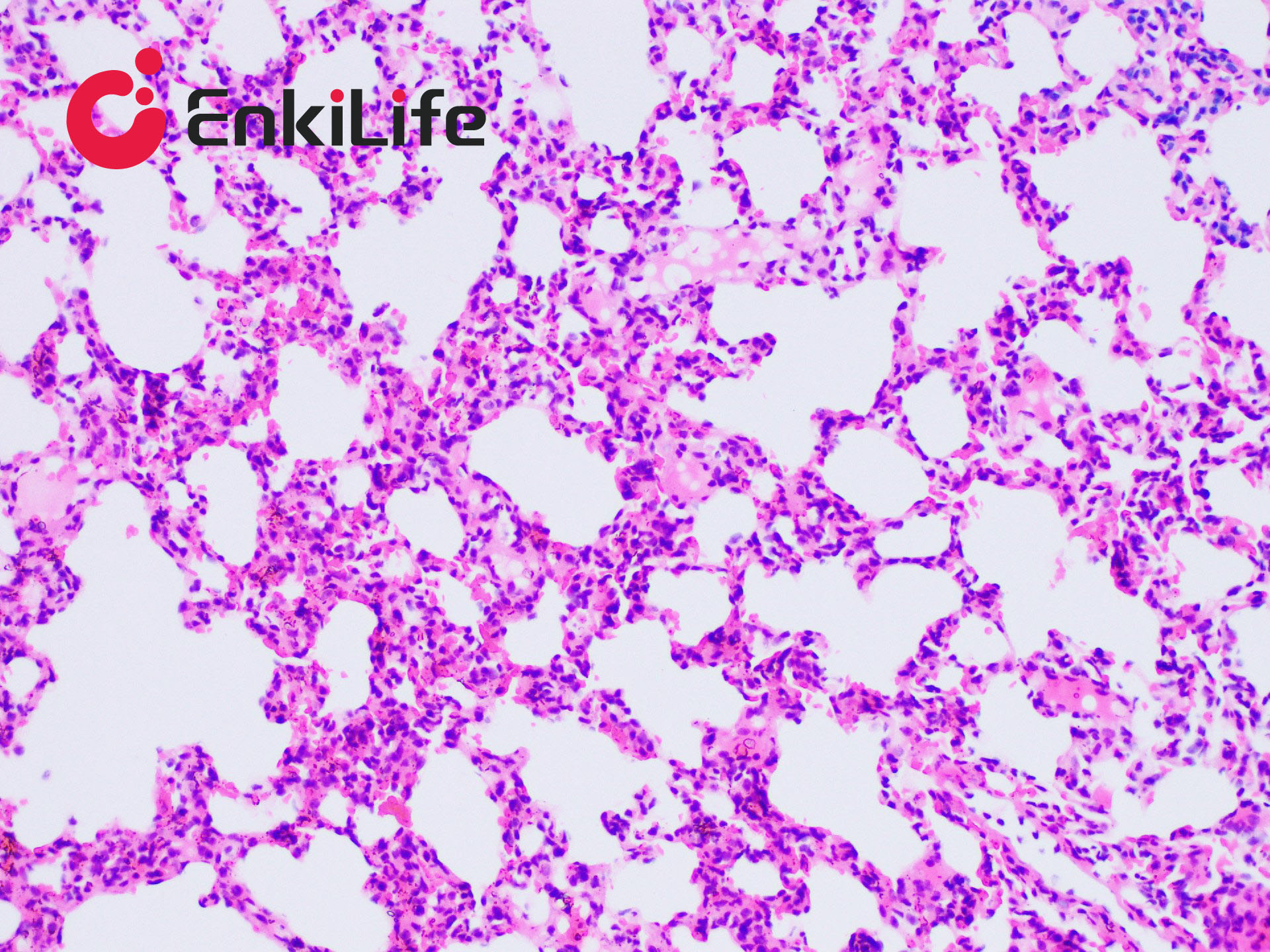
Glycogen staining is one of the routine procedures in pathology. In 1946 McManus first applied the periodic acid–Schiff (PAS) technique to demonstrate mucins. The method is commonly used to visualize glycogen and other polysaccharides, and it additionally reveals neutral mucosubstances and certain acidic materials. Combined Alcian Blue (also known as Alcian Blue 8GX or Ingrain Blue 1) and PAS staining permits the simultaneous identification of neutral and acidic mucins in the same tissue section. The protocol is widely employed as a comprehensive mucin-detection tool: sections are first stained with standard Alcian Blue (pH 2.5), followed by the PAS reaction. Alcian Blue stains sialomucins, sulfomucins and proteoglycans blue, whereas the PAS reaction imparts a deep red/magenta color to neutral mucins. Tissues and cells that contain both neutral and acidic mucins appear in various shades of purple because Alcian Blue and Schiff reagent can interact. A typical example is the goblet cells of the small intestine, which contain both neutral and sialomucins. The staining mechanism of Alcian Blue relies on its being a copper-containing phthalocyanin dye that carries a cationic charge. The dye forms insoluble complexes with anionic groups (carboxyl and sulfate) in tissue mucins. Positively charged salt linkages between the dye and negatively charged acidic groups of acid mucopolysaccharides produce the blue color. Subsequent PAS staining then allows three different mucosubstance components to be distinguished.
EnkiLife Alcian Blue-Periodic Acid-Schiff (AB-PAS) Staining Solution contains Alcian Blue staining solution and Schiff reagent as core components. After the procedure, glycogen, neutral mucins and various glycoproteins appear magenta, while acidic mucins remain blue. The kit is intended for research use only and is not suitable for clinical diagnosis or any other application.
Basic Information
Product name | Alcian Blue-Periodic Acid-Schiff (AB-PAS) Staining Kit |
Sizes | 50 mL, 100 mL |
Storage | 2-8 °C,keep away from light |
Shipping | Shipped with ice pack |
Validity | 12 months |
Product Components
Components | 6x 50mL | 6x 100mL |
Reagent (A): Alcian Blue Staining Solution | 50 mL | 100 mL |
Reagent (B): Periodic Acid Solution | 50 mL | 100 mL |
Reagent (C): Schiff Reagent | 50 mL | 100 mL |
Reagent (D): Hematoxylin Staining Solution | 50 mL | 100 mL |
Reagent (E): Acid Differentiation Solution | 50 mL | 100 mL |
Reagent (F): Scott Bluing Solution | 50 mL | 100 mL |
Notes
1. Sections must be completely dewaxed; residual paraffin will impair staining.
2. Do not prolong oxidation in periodic acid; optimal temperature is 18–22 °C.
3. Alcian Blue Staining Solution, Periodic Acid Solution and Schiff Reagent must be kept tightly closed at 4 °C and protected from light and air. Allow reagents to equilibrate to room temperature for ~30 min before use and perform incubations in the dark.
4. Replace Acid Differentiation Solution regularly. Differentiation time depends on section thickness, tissue type and reagent age; rinse thoroughly in tap water afterwards.
5. Incubation times in Periodic Acid Solution and Schiff Reagent are critical and should be adjusted according to section thickness and tissue type.
6. When counterstaining nuclei with Hematoxylin Staining Solution, keep the staining light to avoid masking the Alcian Blue signal.
7. The order of AB and PAS steps influences the final color: if PAS is performed before Alcian Blue, neutral mucins and glycogen may appear purple; if Alcian Blue is performed first, these components will be the expected magenta.
8. Wear laboratory coat and disposable gloves for personal safety.
9. Use reagents promptly after opening to ensure consistent results.